Anterograde transport of rabies virus in DRG neurons.
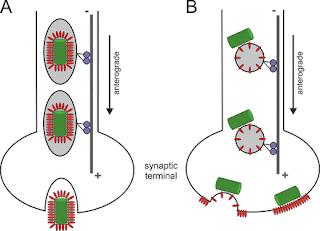 |
| Model of intraneuronal postreplicative RABV transport. |
Rabies virus (RABV) spread is widely accepted to occur only by retrograde axonal transport. However, examples of anterograde RABV spread in peripheral neurons such as dorsal root ganglion (DRG) neurons indicated a possible bidirectional transport by an uncharacterized mechanism. Here, we analyzed the axonal transport of fluorescence-labeled RABV in DRG neurons by live-cell microscopy. Both entry-related retrograde transport of RABV after infection at axon endings and postreplicative transport of newly formed virus were visualized in compartmentalized DRG neuron cultures. Whereas entry-related transport at 1.5 μm/s occurred only retrogradely, after 2 days of infection, multiple particles were observed in axons moving in both the anterograde and retrograde directions. The dynamics of postreplicative retrograde transport (1.6 μm/s) were similar to those of entry-related retrograde transport. In contrast, anterograde particle transport at 3.4 μm/s was faster, indicating active particle transport. Interestingly, RABV missing the glycoproteins did not move anterogradely within the axon. Thus, anterograde RABV particle transport depended on the RABV glycoprotein. Moreover, colocalization of green fluorescent protein (GFP)-labeled ribonucleoproteins (RNPs) and glycoprotein in distal axonal regions as well as cotransport of labeled RNPs with membrane-anchored mCherry reporter confirmed that either complete enveloped virus particles or vesicle associated RNPs were transported. Our data show that anterograde RABV movement in peripheral DRG neurons occurs by active motor protein-dependent transport. We propose two models for postreplicative long-distance transport in peripheral neurons: either transport of complete virus particles or cotransport of RNPs and G-containing vesicles through axons to release virus at distal sites of infected DRG neurons.
REFERENCE:
Bauer A, et al. Anterograde glycoprotein-dependent transport of newly generated rabies virus in dorsal root ganglion neurons. J Virol. 2014 Dec;88(24):14172-83. doi: 10.1128/JVI.02254-14. Epub 2014 Oct 1. PubMed PMID: 25275124.

Comentarios
Publicar un comentario